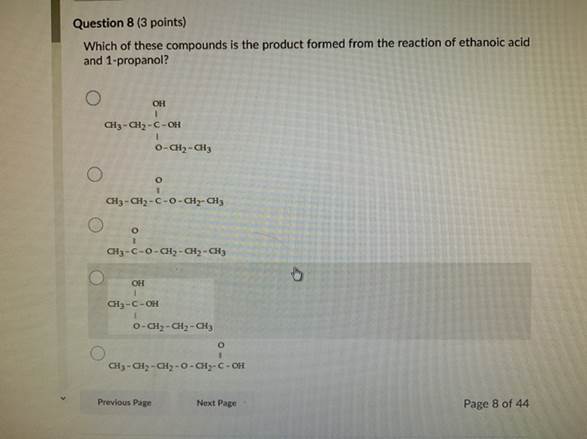
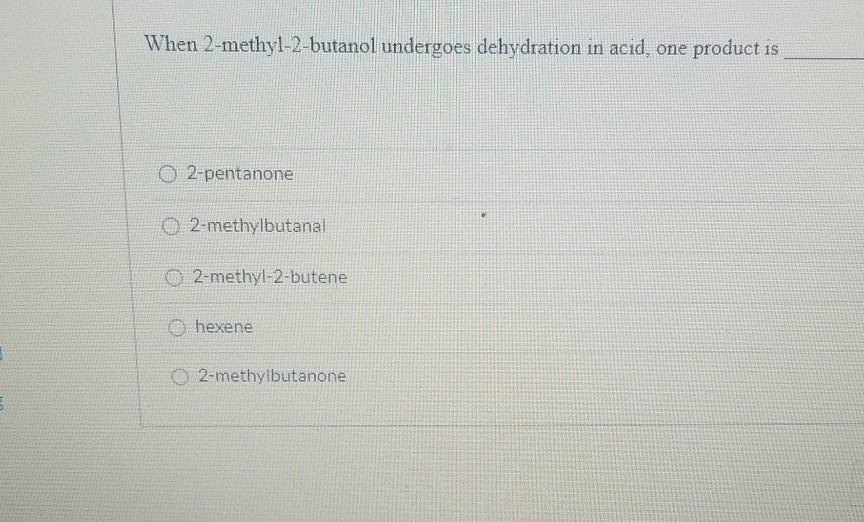
Dehydration Of 2 Methyl 2 Butanol Mechanism
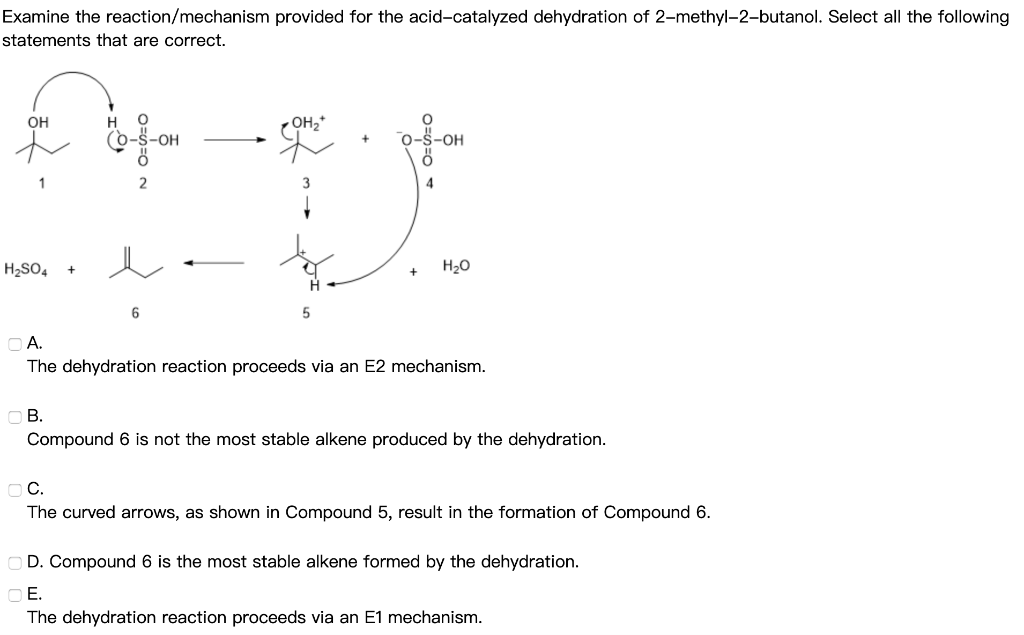
Alright, buckle up, science adventurers! Today, we're diving headfirst into a little chemical shindig that’s surprisingly fun – the dehydration of 2-methyl-2-butanol. Now, I know what you're thinking: "Dehydration? Sounds like a terrible party!" But trust me, this is the kind of dehydration that makes things more exciting, not less. Think of it as a chemical spa day for our friend, 2-methyl-2-butanol, where it sheds a little something to become something even cooler.
So, who is this mysterious 2-methyl-2-butanol? Imagine a tiny, branched party animal. It’s an alcohol, so it’s got that little -OH group hanging around, like a friendly bartender always ready to mix things up. Its name tells us a lot: a four-carbon chain (that's the 'butanol' part), a methyl group (a single carbon friend) hanging off the second carbon, and the alcohol group also chilling on that second carbon. It’s quite the social butterfly, this one!
Now, what does "dehydration" mean in our chemical world? It's basically like a chemical's way of saying, "Excuse me, I'm going to lose a water molecule." Yep, just like you might lose a little sweat on a hot day, our 2-methyl-2-butanol is going to ditch a water molecule. And when it does, oh boy, does it transform!
Must Read
To make this happen, we need a little help. Think of it like needing a really good DJ to get the party started. In our case, the DJ is a strong acid, like sulfuric acid or phosphoric acid. These acids are like the super-enthusiastic party organizers, pushing everyone to get into the groove. They’re not actually part of the final product, but they’re essential for kicking off the whole transformation. They’re the hype men of the chemical world!
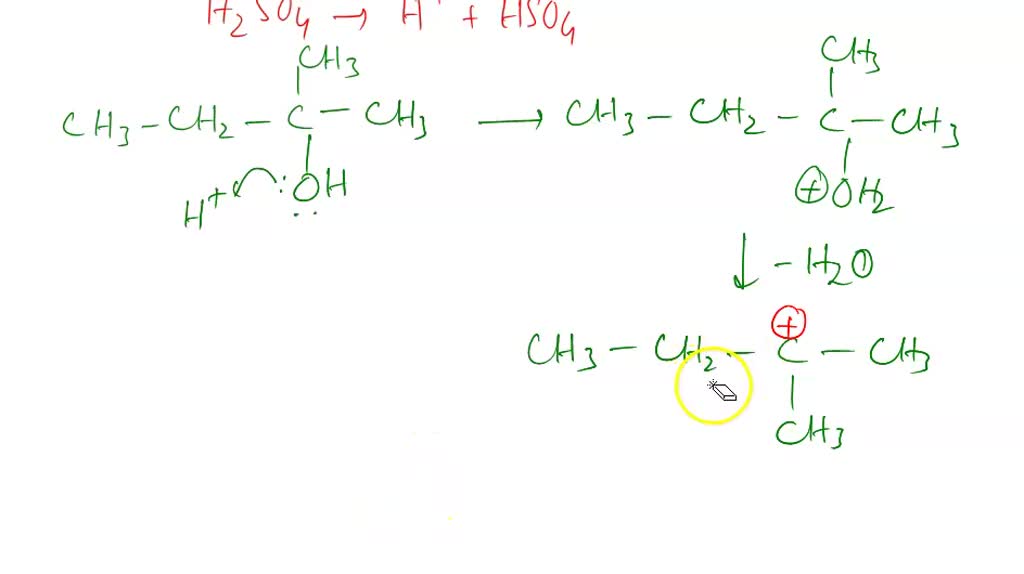
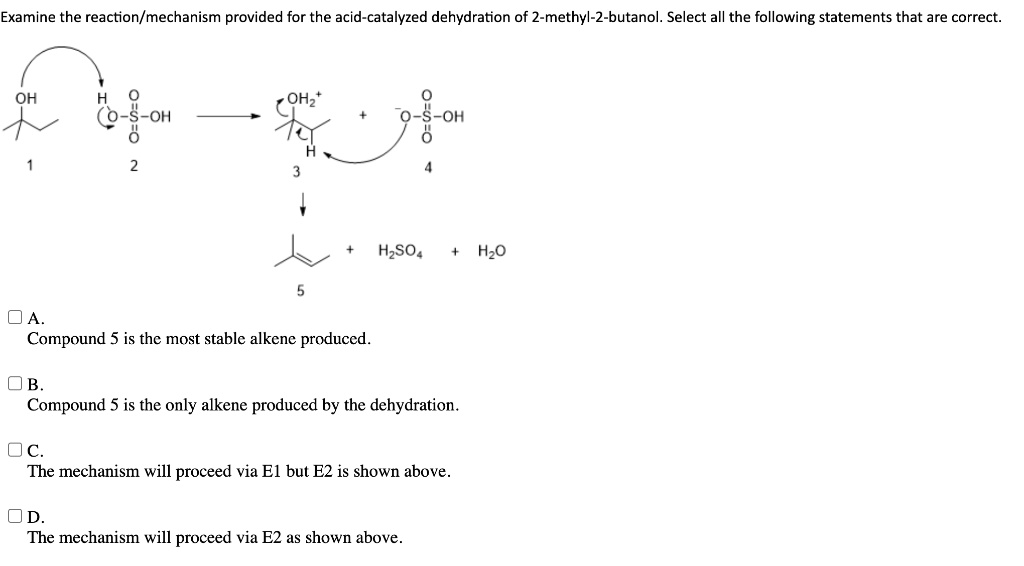
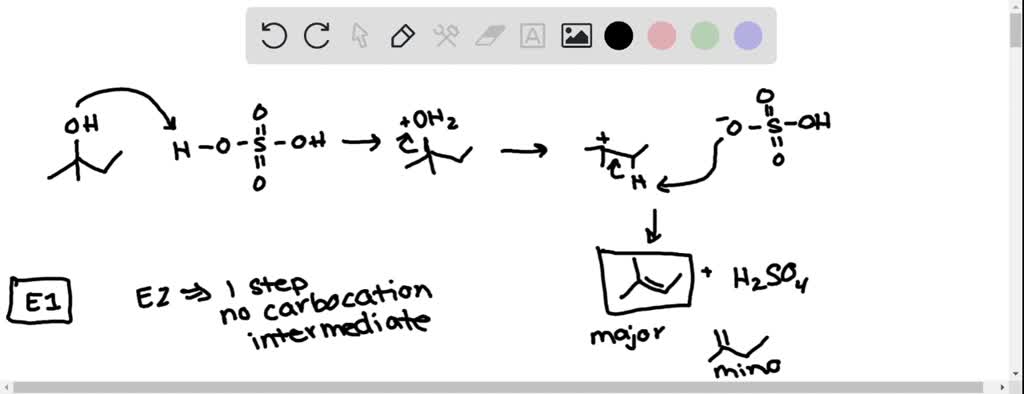
So, here’s where the magic really begins. Our acid DJ arrives, sees 2-methyl-2-butanol, and whispers, "Hey, you look a little heavy with that -OH group. Let's lighten you up!" The acid then latches onto the -OH group, making it an even better leaving group. Think of it as the acid giving the -OH group a little pep talk: "Go on, you can do it! You're destined for greater things... like becoming part of a water molecule!"
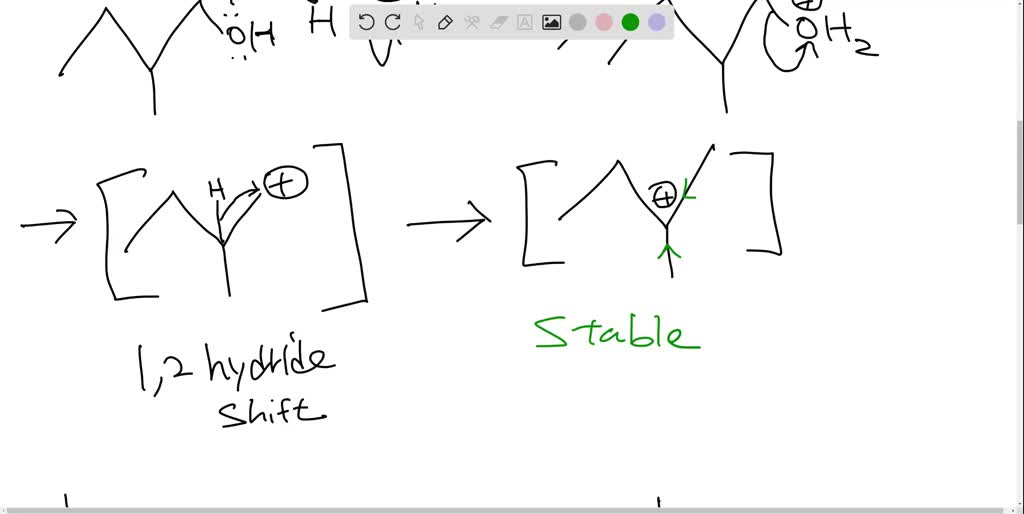
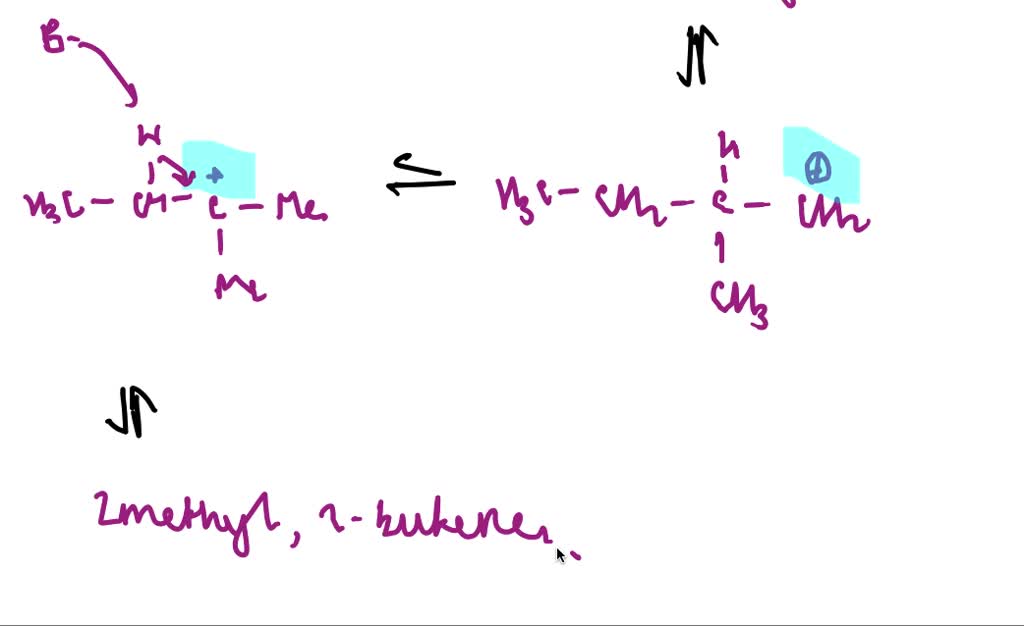
Once that -OH group is all jazzed up by the acid, it’s ready to bounce. It peaces out, taking a hydrogen atom from a neighboring carbon atom with it. And this is where the truly spectacular stuff happens. As the -OH and the hydrogen atom say their goodbyes (forming a happy little H2O molecule, off to enjoy its own independent life), the two carbon atoms they left behind realize they’ve got some extra 'hands' (electrons) to hold. And what do two carbon atoms do when they have extra hands? They get really close and form a double bond! It’s like two friends high-fiving so hard they form a permanent handshake. Bam! A double bond is born!
This double bond is the signature of our new creation. Our 2-methyl-2-butanol, the branched party animal, has now shed its water and become an alkene. Specifically, it’s going to form 2-methylbut-2-ene. Look at that! It's still got that four-carbon backbone, and the methyl group is still there, but now there’s a super-strong double bond between the second and third carbons. It’s like our friend went from a casual stroll to a full-on sprint! It’s more reactive, more energetic, ready for its next adventure.
Imagine 2-methyl-2-butanol as a person at a dance party. They’re having a good time, but they’re a little hesitant. The acid comes along, like a charismatic friend, and says, "Come on, let's really move!" The person feels a surge of energy, and suddenly, they’re doing this amazing spin (forming the double bond) and leaving behind a little bit of sweat (the water molecule). Now they're the life of the party!
The beauty of this reaction is that because our starting material, 2-methyl-2-butanol, is a tertiary alcohol (meaning the carbon attached to the -OH group is connected to three other carbons), it’s perfectly set up for this kind of "carbocation" shuffle. When the -OH group leaves, it leaves behind a positively charged carbon atom – a carbocation. This little guy is super eager to stabilize, and it’s perfectly positioned to grab a hydrogen from the adjacent carbon to form that glorious double bond. It’s like the universe saying, "Yep, everything’s in the right place for some serious bonding!"
There might even be a slight temptation for other possible alkenes to form, like 2-methylbut-1-ene. But our friend, 2-methyl-2-butanol, is a bit of a perfectionist when it comes to forming that double bond. It’s going to preferentially form the most stable alkene, and in this case, that’s the one with the double bond sitting right in the middle, surrounded by lots of carbon friends. It’s like choosing the most popular spot on the dance floor!
So, there you have it! The dehydration of 2-methyl-2-butanol is a fantastic little chemical transformation. It's a story of shedding the old to embrace the new, of a little push from an acid friend leading to the formation of a vibrant double bond. It’s proof that sometimes, losing a little bit of yourself can lead to becoming something even more exciting and dynamic. Who knew chemistry could be so… liberating?