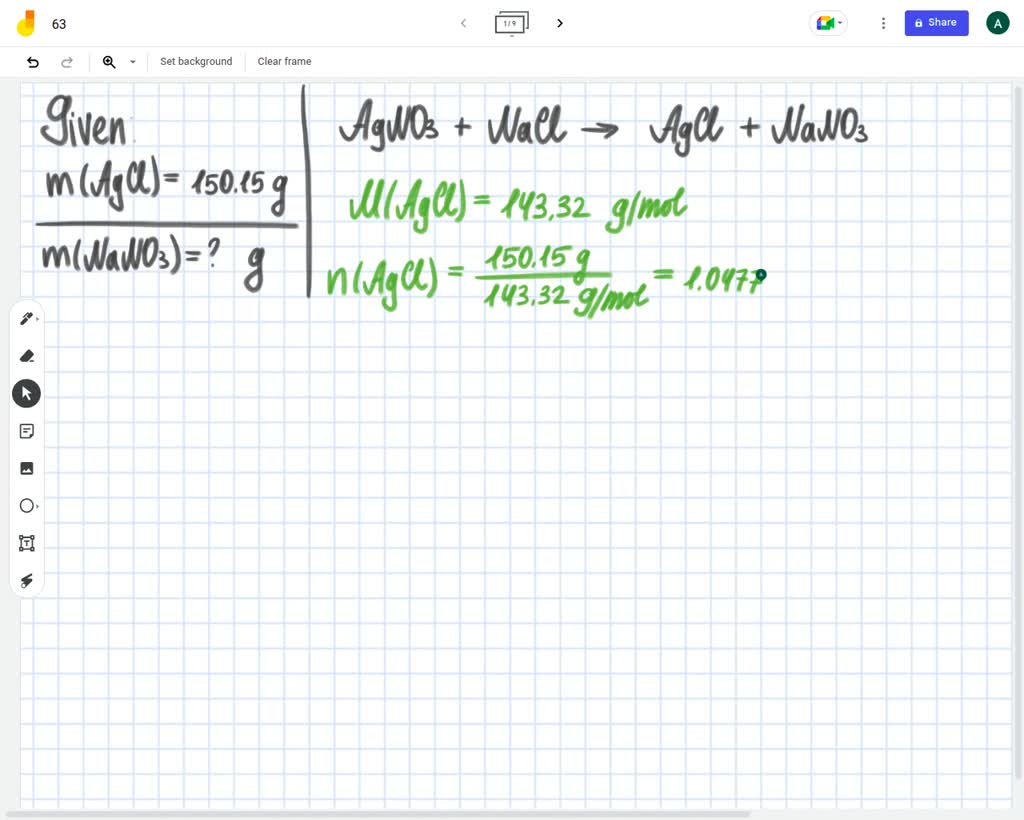
What Is The Mass Of Silver In 3.4 G Agno3
Hey there, fellow explorers of the everyday! Ever find yourself staring at a little pile of something interesting – maybe a bit of sparkle, a new gadget part, or even a mystery ingredient from a DIY project – and a little voice pops into your head asking, "But what's in this?" Yeah, we've all been there. It’s that curious itch, the desire to know the nitty-gritty behind the stuff that populates our lives. Today, we're diving into a question that might sound a tad scientific, but trust us, it’s got a surprisingly chill vibe. We're talking about the humble, yet fascinating, compound known as silver nitrate, or AgNO₃ for those in the know. And specifically, we’re going to unravel a little puzzle: what’s the mass of silver tucked away inside a mere 3.4 grams of AgNO₃? Let's make this as easy-going as a Sunday morning coffee, shall we?
Think of it like this: you’ve got a delightful little bag of fancy jellybeans, all mixed up. You know the bag weighs a certain amount, but you’re curious about how many of those jellybeans are your absolute favorite cherry flavor. That’s kind of what we’re doing with silver nitrate. It’s a compound, a blend of different elements, and we want to isolate the silver component’s weight. No need for a lab coat or a stern lecture from Mr. Henderson (remember him from high school chem?). We're keeping it light, breezy, and dare we say, a little bit chic.
So, what exactly is silver nitrate? It’s that classic white crystalline solid that has some seriously cool applications. Back in the day, it was a go-to for photography – those old black and white photos? A lot of that magic involved silver nitrate’s sensitivity to light. Pretty neat, right? It’s also used in some medical applications, like treating certain skin conditions or as an antiseptic. Think of it as a tiny, potent powerhouse. It’s got that sparkle, but it also has a purpose. And that purpose is often tied to the silver it contains.
Must Read
Now, before we get our hands dirty with numbers (don't worry, they're friendly numbers!), let's briefly touch on why understanding the composition of compounds matters. It’s not just for scientists in white coats. In our everyday lives, it helps us make informed choices. Whether you're looking at nutritional labels on your favorite snacks, understanding the ingredients in your skincare, or even appreciating the metallurgy behind your favorite jewelry, knowing what’s what is empowering. It's like being able to read the secret code of the world around you. And for this particular code, the key lies in something called molar mass.
Don't let the term "molar mass" scare you off! It's simply the weight of one mole of a substance. A mole is just a giant, convenient counting unit for atoms and molecules – think of it as a baker's dozen, but for microscopic particles. So, the molar mass of silver nitrate tells us the total weight of one mole of AgNO₃. To figure this out, we need the atomic masses of its constituent elements: silver (Ag), nitrogen (N), and oxygen (O).
Here’s where our trusty periodic table comes into play. You might have one hanging in a dusty classroom somewhere, or maybe you've seen it online. For our purposes, let's snag the approximate atomic masses: * Silver (Ag): ~107.87 g/mol * Nitrogen (N): ~14.01 g/mol * Oxygen (O): ~16.00 g/mol
Since our silver nitrate molecule (AgNO₃) has one silver atom, one nitrogen atom, and three oxygen atoms, we can calculate its total molar mass. It’s like assembling a LEGO structure – you add up the weights of all the individual bricks. So, Molar Mass of AgNO₃ = (1 × Atomic Mass of Ag) + (1 × Atomic Mass of N) + (3 × Atomic Mass of O) Molar Mass of AgNO₃ ≈ (1 × 107.87) + (1 × 14.01) + (3 × 16.00) Molar Mass of AgNO₃ ≈ 107.87 + 14.01 + 48.00 Molar Mass of AgNO₃ ≈ 169.88 g/mol

There we have it! One mole of silver nitrate weighs approximately 169.88 grams. This number is our gateway to understanding the silver content. It’s the recipe for our compound, telling us the proportion of each element by weight.
Now, we're interested in the mass of silver within our specific 3.4 grams of AgNO₃. We can use a simple proportion or a percentage. The percentage of silver in silver nitrate is the mass of silver divided by the total molar mass of silver nitrate, multiplied by 100. Percentage of Ag = (Mass of Ag / Molar Mass of AgNO₃) × 100 Percentage of Ag ≈ (107.87 g/mol / 169.88 g/mol) × 100 Percentage of Ag ≈ 0.6350 × 100 Percentage of Ag ≈ 63.50%
So, roughly 63.50% of silver nitrate is actually silver! This is a pretty significant chunk. It means that for every 100 grams of silver nitrate you have, you have about 63.5 grams of pure silver. Pretty cool, right? It’s like finding out that your favorite cookie is mostly chocolate chips! The sweetness of discovery!
Now, let's apply this percentage to our specific 3.4 grams of AgNO₃. We just need to take that percentage and multiply it by our total mass.
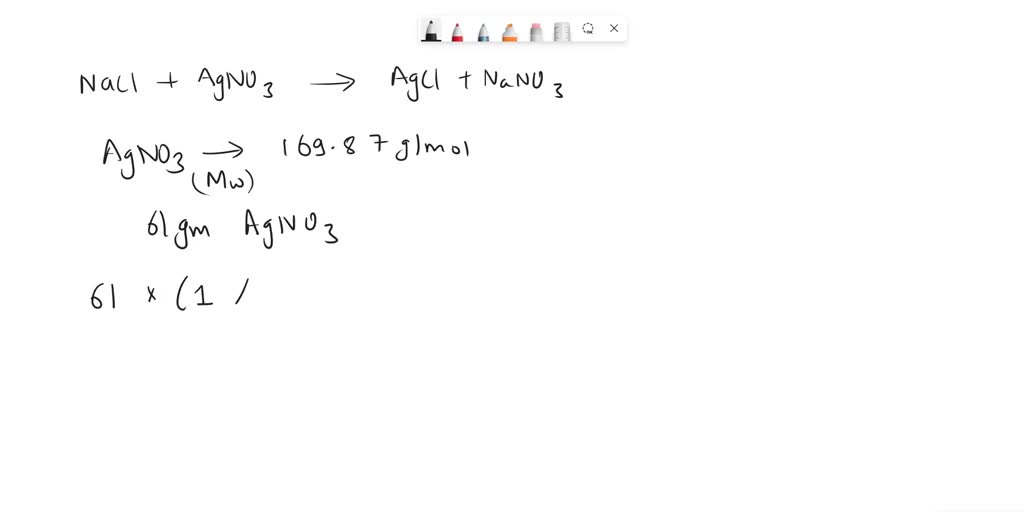
Mass of Silver in 3.4 g AgNO₃ = Total Mass of AgNO₃ × Percentage of Ag
Mass of Silver in 3.4 g AgNO₃ = 3.4 g × 0.6350
Mass of Silver in 3.4 g AgNO₃ ≈ 2.16 grams
And there you have it! In 3.4 grams of silver nitrate, there are approximately 2.16 grams of silver. See? Not so intimidating after all. It’s like breaking down a complex recipe into simple, manageable steps. You start with the ingredients (atomic masses), combine them according to the instructions (molar mass calculation), and then figure out the proportion of your favorite ingredient (percentage of silver).
Let's take a moment to appreciate this. Silver, as a precious metal, has been prized for millennia. It’s used in everything from elegant silverware and intricate jewelry to essential components in electronics. Knowing that a small amount of a compound like silver nitrate contains such a substantial proportion of this valuable element gives you a little peek into the hidden riches that surround us. It’s like finding a secret stash of treasure in your pantry!
Fun Little Fact Break! Did you know that the ancient Romans used silver coins not just for their value, but also to keep their wine from spoiling? They’d drop silver coins into amphorae of wine, and the silver ions released helped inhibit bacterial growth. Talk about a practical, ancient life hack! It’s a testament to silver’s enduring utility, even if its primary use now isn't preserving your Pinot Grigio.
Now, let’s sprinkle in some practical tips. Where might you encounter silver nitrate, or a similar compound, in your daily life? Beyond the historical photography angle, it’s sometimes found in wart removal treatments (yes, really!) or in certain types of mirrors. If you’re dabbling in DIY, especially anything involving chemistry sets or specific art materials, you might come across it. And if you’re ever curious about the composition of something you own, a quick online search using the chemical formula can often lead you to resources that explain its makeup, much like we’ve done today.
Cultural Nod! Silver has been a symbol of wealth, purity, and prosperity across many cultures for centuries. From the intricate silver filigree found in traditional Indian jewelry to the elegant silver candelabra gracing a Hanukkah menorah, silver is woven into the fabric of human history and celebration. It’s more than just a metal; it’s a storyteller.

When you’re handling anything that involves chemical compounds, even if it’s just for curiosity’s sake, a little bit of caution goes a long way. Always follow any safety instructions provided, work in a well-ventilated area, and if you’re unsure about something, it's always best to consult an expert or err on the side of caution. Think of it as being a responsible curator of your own little scientific discoveries.
The beauty of these calculations is that they can be applied to so many things. If you’re interested in the gold content in a piece of jewelry, the iron in your multivitamin, or the calcium in your milk, the principles are often the same: understand the compound, know the atomic masses, and calculate the proportions. It’s a way of demystifying the world and appreciating the intricate dance of elements that make up everything we see and touch.
Let’s recap our little adventure. We started with 3.4 grams of silver nitrate (AgNO₃) and, using a bit of chemical sleuthing, discovered that approximately 2.16 grams of that is pure, shimmering silver. We learned about molar mass, the importance of atomic masses, and how to calculate percentages within a compound. We also took detours through the history of photography, the practical uses of silver, and its cultural significance. All from a simple question!
A Moment of Reflection: In our fast-paced world, it’s easy to overlook the intricate details. We use things, we consume things, and we often don’t pause to consider what they’re truly made of. But taking a moment to understand the composition of even the smallest things, like a few grams of silver nitrate, can foster a deeper appreciation for the world around us. It reminds us that everything, from the simplest compound to the most complex organism, is a marvel of elemental interactions. It’s a gentle nudge to be a little more curious, a little more observant, and a little more in tune with the beautiful chemistry of life itself. So, the next time you’re holding something, anything at all, give it a curious glance. You might just discover a hidden story, a forgotten application, or a surprising abundance of a valuable element, just waiting to be appreciated.

![[ANSWERED] AlCl3 AgNO3 Al NO3 AgCl If 30 7 g of aluminum chloride is](https://media.kunduz.com/media/sug-question-candidate/20210527170714164561-3285608.jpg?h=512)